What Is Modafinil and What Is It Used For?
Drug Class: Modafinil is a prescription central nervous system (CNS) wake-promoting agent. It is a Schedule IV controlled substance in the United States.
Approved Medical Uses
The FDA has approved modafinil to improve wakefulness in adults with excessive sleepiness linked to 3 specific conditions:
- Narcolepsy — a sleep disorder that causes uncontrollable daytime sleepiness. Modafinil helps with daytime fatigue but has not been shown to improve cataplexy (sudden muscle weakness).
- Obstructive sleep apnea (OSA) — modafinil treats the excessive sleepiness caused by OSA. It does not treat the underlying airway obstruction itself.
- Shift work disorder (SWD) — excessive sleepiness in people who work non-traditional hours (nights, rotating shifts).
How Modafinil Works
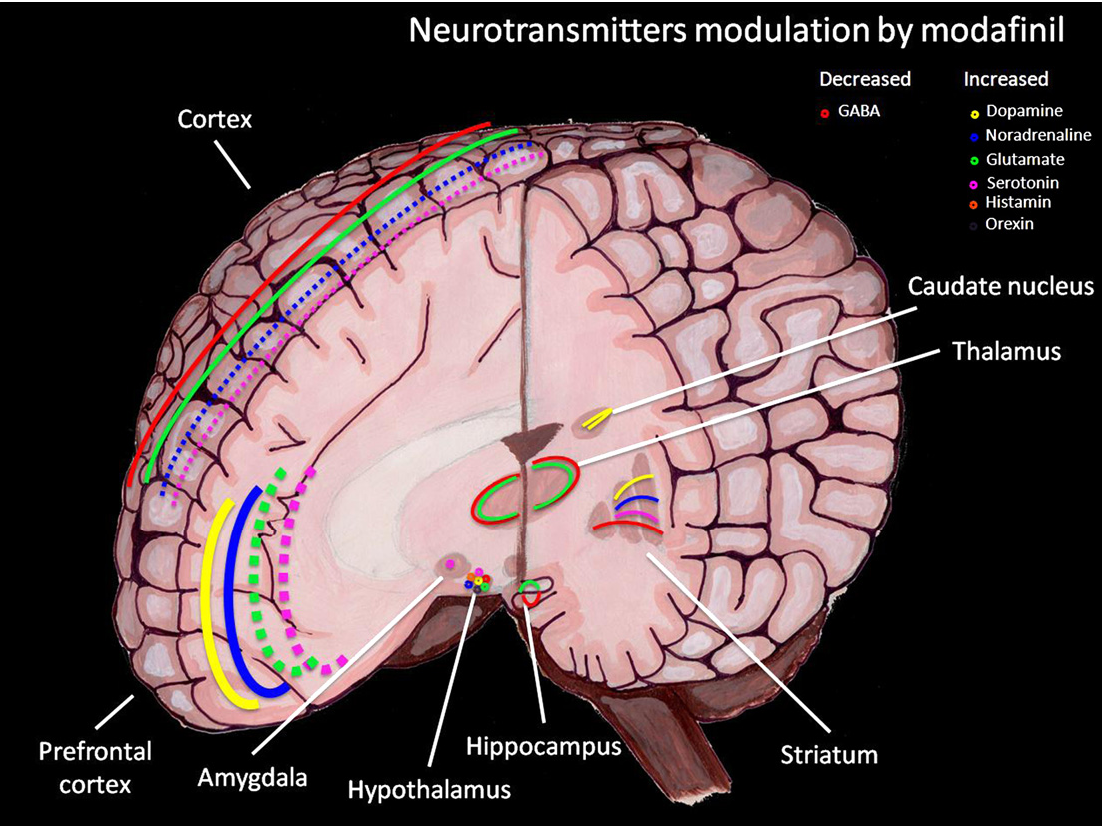
The exact way modafinil promotes wakefulness is not fully understood. According to the FDA label, its precise mechanism is unknown.
- It has wake-promoting effects similar to stimulants like amphetamine and methylphenidate — but its pharmacologic profile is not identical to those drugs.
- Research suggests it most likely works by enhancing norepinephrine and dopamine activity in brain areas that control wakefulness.
- Because it acts differently from traditional stimulants, it is generally considered to have a lower potential for abuse — though it remains a controlled substance.
Note: Modafinil treats sleepiness symptoms only. It is not a substitute for sleep and does not treat the root cause of any underlying sleep disorder.
Modafinil Dosage: How Much Should You Take?
Condition-Specific Dosing
- Narcolepsy and obstructive sleep apnea (OSA): 200 mg taken by mouth once a day as a single morning dose.
- Shift work disorder (SWD): 200 mg taken by mouth once a day, approximately 1 hour before the start of your work shift.
Note: Doses up to 400 mg/day as a single dose have been well tolerated for narcolepsy and OSA. However, there is no consistent evidence that 400 mg works better than 200 mg.
Missed Dose and Storage
If you miss a dose:
Take the missed dose as soon as you remember. However, if it is close to your normal bedtime, skip the missed dose to avoid interference with your sleep. Never take a double dose to make up for a missed one.
Storage:
Store Modafinil at room temperature, between 20°C to 25°C (68°F to 77°F). As a Schedule IV controlled substance, Modafinil must be kept in a secure place to prevent misuse or diversion.
- If you remember a missed dose, take it as soon as possible — however, do not take it if you do not plan to be awake for several hours, as Modafinil has a long half-life (approximately 12–15 hours) and late-day administration can cause significant insomnia or sleep disturbances.
- If it is close to bedtime, skip the missed dose entirely and take your next dose at the regular time.
- Go back to your regular dosing schedule. For patients with Shift Work Disorder (SWD), remember that your dose should be taken approximately 1 hour prior to the start of your work shift; consult your healthcare provider if you are unsure how to manage a missed dose.
Important: Never take 2 doses at once to make up for a missed one. Taking double doses does not improve results and may increase your risk of side effects.
Modafinil Side Effects and Safety Warnings
Important Safety Warning: Modafinil can cause serious, life-threatening skin reactions and psychiatric effects. Stop taking modafinil and get emergency medical help immediately if you develop a rash, blistering, peeling skin, or any new or worsening mental health symptoms.
Serious and Life-Threatening Reactions
Modafinil has been linked to severe reactions that require immediate medical attention. These are not common, but they can be life-threatening.
Stop taking modafinil at the first sign of a rash — unless your doctor confirms it is clearly not related to the drug. Serious skin reactions reported with modafinil include:
- Stevens-Johnson Syndrome (SJS) — a severe skin and mucous membrane reaction
- Toxic Epidermal Necrolysis (TEN) — widespread skin peeling that can be fatal
- Drug Rash with Eosinophilia and Systemic Symptoms (DRESS) — a reaction affecting the skin and internal organs
These reactions have been reported after modafinil use and may require hospitalization.
Modafinil can cause serious mental health reactions, including:
- Mania (extreme elevated mood or energy)
- Delusions and hallucinations
- Suicidal thoughts
- Aggression
Some of these reactions have led to hospitalization. Patients with a history of psychosis, depression, or mania should use modafinil with caution. If any psychiatric symptoms develop, talk to your doctor right away — stopping modafinil may be necessary.
Note: If you have a history of mental health conditions, make sure your prescribing doctor is aware before starting modafinil.
Contraindications, Precautions, and Drug Interactions
Important Safety Warning: Modafinil can cause serious skin reactions (including Stevens-Johnson syndrome), severe allergic reactions, and multi-organ hypersensitivity. Seek emergency care immediately if you develop a rash, swelling, or difficulty breathing while taking this medication.
Who Should Not Take Modafinil
Talk to your doctor before taking modafinil if any of the following apply to you:
- Known allergy to modafinil or armodafinil, or any inactive ingredients in the tablet (absolute contraindication)
- Heart conditions (warnings and precautions): history of left ventricular hypertrophy (thickening of the heart wall), or mitral valve prolapse syndrome previously experienced while taking CNS stimulants; modafinil is not recommended in these patients. Caution is also advised in patients with unstable angina, recent heart attack, or hypertension, as modafinil can increase blood pressure and heart rate and monitoring is required.
- Psychiatric history (warnings and precautions): past episodes of psychosis, depression, or mania; modafinil should be used with caution due to the risk of psychiatric adverse events
- Severe liver impairment: your doctor may need to reduce your dose by half
Key Drug and Substance Interactions
Warning: Birth Control Failure Risk. Modafinil reduces the effectiveness of hormonal birth control (such as pills, patches, or rings containing ethinyl estradiol). This effect lasts for 1 month after you stop taking modafinil. Use a backup or alternative contraceptive method during treatment and for 1 full month afterward.
| Interaction | What Happens |
|---|---|
| Hormonal contraceptives (e.g., ethinyl estradiol) | Modafinil is a moderate CYP3A4/5 inducer that speeds up the breakdown of steroidal contraceptives, reducing their effectiveness. Although the AUC of ethinyl estradiol is decreased by approximately 18%, this figure alone does not capture the full clinical risk of contraceptive failure. Hormonal contraceptives — including pills, patches, and implants — may be unreliable during treatment; an alternative or additional method of contraception is required during therapy and for one month after stopping modafinil. Non-hormonal options (e.g., copper IUDs) or methods less dependent on systemic CYP3A4 metabolism (e.g., hormonal IUDs, which act primarily through local mechanisms) are generally considered reliable alternatives. |
| Armodafinil (a related drug) | Also reduces hormonal contraceptive effectiveness by lowering systemic exposure to ethinyl estradiol; notably, the AUC of ethinyl estradiol is decreased by approximately 24% with armodafinil — a somewhat greater reduction than the 18% seen with modafinil. The same precaution applies: use an alternative or concomitant contraceptive method during treatment and for one month after discontinuation. |
Modafinil Cost and How to Save Money
Retail Price vs. Generic Cost
Modafinil can be expensive, especially without insurance coverage.
| Option | Estimated Cost |
|---|---|
| Brand-name modafinil/Provigil (30 tablets, 100 mg) | ~$1,600–$1,800 (WAC) |
| Generic modafinil (30 tablets, 100 mg) | ~$10–$45 (with discount card/coupon) |
| Average wholesale acquisition cost per tablet (100 mg) | ~$0.25–$0.40 (NADAC) |
Note: These prices are estimates as of April 2026 and are subject to change.
Getting insurance to cover modafinil can be difficult, depending on your plan.
Tips to Lower Your Modafinil Cost
- Choose generic: Generic modafinil has been available since June 2012 and costs significantly less than the brand-name version.
- Use a drug price comparison tool: Services like drug discount websites provide real-time pharmacy pricing and coupons that can lower your out-of-pocket cost.
- Compare pharmacies: Prices vary by location — checking multiple pharmacies can lead to meaningful savings.
Note: Always confirm current pricing directly with your pharmacy, as costs change frequently.
Frequently Asked Questions
What conditions is modafinil approved to treat?
Modafinil (brand name Provigil) is FDA-approved to improve wakefulness in adults with excessive sleepiness caused by 3 conditions:
- Narcolepsy
- Obstructive sleep apnea (OSA) — for the sleepiness only, not the underlying airway obstruction
- Shift work disorder (SWD)
Can modafinil affect my birth control?
Important Safety Warning: Yes. Modafinil can reduce how well hormonal birth control works — during treatment and for 1 month after stopping. Talk to your doctor about using an additional or different method of contraception during this time.
Is it safe to take modafinil during pregnancy or while breastfeeding?
Important Safety Warning: Modafinil may cause fetal harm based on animal data and human pregnancy studies. It should not be used during pregnancy unless a doctor determines the benefit clearly outweighs the risk to the baby.
Modafinil is not considered safe during pregnancy. Multiple studies have linked it to a higher rate of birth defects (congenital malformations) compared to the general population. Patients are advised to avoid modafinil while pregnant and to discuss safer alternatives with their doctor.
Pregnancy Risks
- Animal studies show modafinil can harm a developing fetus, including embryotoxicity (toxic effects on the embryo) even without harm to the mother.
- A Danish study found a 12% rate of birth defects in babies exposed to modafinil in the first trimester, compared to 3.9% in unexposed babies.
- A U.S. Pregnancy Registry analysis of 102 live births found 13% had major birth defects — well above the general population rate of about 3%. Defects included heart problems, hypospadias (a urinary tract malformation), and congenital torticollis (a neck muscle condition).
- Intrauterine growth restriction (poor fetal growth) and spontaneous abortion (miscarriage) have also been reported with modafinil use.
Breastfeeding
The picture is less clear for breastfeeding. The official drug label notes it is not fully known whether modafinil passes into breast milk. However:
- Some case reports and pharmacokinetic estimates (e.g., a relative infant dose of approximately 5.3%, below the commonly cited 10% threshold of concern) exist in the literature; however, modafinil and its metabolites may be present in human milk, and the safety profile for the breastfed infant has not been established — the FDA-approved labeling explicitly states that caution should be exercised.
- More critically, modafinil may cause fetal harm when administered during pregnancy: data from the 2024 FDA labeling and pregnancy registries indicate an increased risk of major congenital malformations, including congenital cardiac anomalies, with a prevalence of approximately 14.7% among exposed pregnancies compared to roughly 3% in the general population. As a result, modafinil is not recommended during pregnancy, and women of childbearing potential are advised to use effective contraception.
Talk to your doctor before using modafinil while breastfeeding. If it is used, the infant should be monitored closely for any side effects.
What is the difference between modafinil 100mg and 200mg?
Both strengths exist, but 200 mg is the standard prescribed dose for all 3 FDA-approved uses. The 100 mg tablet allows for dose adjustments — for example, patients with severe liver problems are advised to take half the normal dose.